MEN vs WOMEN in PCI Outcomes: What’s the Consensus?
Some of us know the data. Some don’t. The question once you do is where we go from here to bridge the gender inequality gap?
To help address this question, SCAI just published in the Journal of the Society for Cardiovascular Angiography & Interventions (JSCAI), the new official journal of SCAI, an “Expert Consensus on Sex Specific Considerations in Myocardial Revascularization." Coronary IVL was referenced in the consensus as a potential first-line therapy for plaque modification of calcified lesions in women specifically.
 JSCAI Publication: Expert Consensus on Sex-Specific Considerations in Myocardial Revascularization
JSCAI Publication: Expert Consensus on Sex-Specific Considerations in Myocardial Revascularization
Read the Publication Here: https://www.jscai.org/article/S2772-9303(21)00016-8/fulltext
The Expert Consensus brings to light the gender disparities in cardiovascular disease, identifies gaps in evidence regarding cardiovascular outcomes in women, inform clinicians on best practices most relevant to female patients and guides future research priorities.
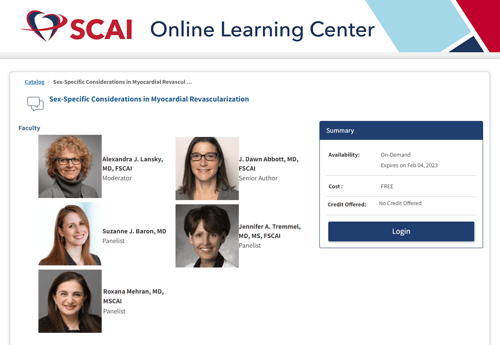
And for more engaging content on this topic, here is a great discussion hosted by SCAI Conversations in Interventional Cardiology with Drs. Alexandra Lansky, J. Dawn Abbott, Jennifer Tremmel, Roxana Mehran, and Suzanne Baron.
SCAI Conversations in Interventional Cardiology Video CME
Sex-Specific Considerations in Myocardial Revascularization (scai.org)
Coronary IVL was referenced in the consensus, stating: “While additional evidence is needed, these results (i.e. Disrupt CAD Pooled Gender Analysis) taken in the context of outcomes with atherectomy devices suggest that IVL may emerge as a first-line therapy for plaque modification of calcified lesions in women specifically.”
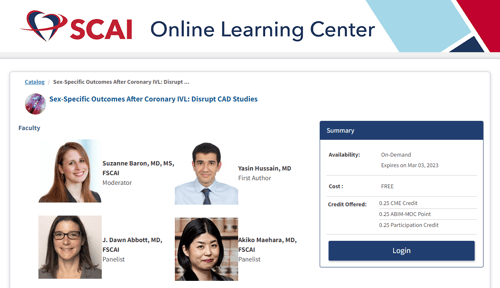
We have included the resources below to learn more about the Disrupt CAD Pooled Gender Analysis and role that coronary IVL plays in calcium modification in men and women.
SCAI Conversations in Interventional Cardiology Video CME
Sex-Specific Outcomes After Coronary IVL: Disrupt CAD Studies (scai.org)
- Plus, the JSCAI CAD Pooled Gender Analysis Publication & Disrupt CAD Pooled Gender Analysis TCT Data Slides.
Be on the lookout, and follow us on Twitter at @ShockwaveIVL, for the updated 1yr outcomes in men vs women from the analysis coming up soon at SCAI’s annual meeting in May 2022.
Important Safety Information
In the United States: Rx only
Indications for Use—The Shockwave Intravascular Lithotripsy (IVL) System with the Shockwave C2 Coronary IVL Catheter is indicated for lithotripsy enabled, low-pressure balloon dilatation of severely calcified, stenotic de novo coronary arteries prior to stenting.
Contraindications—The Shockwave C2 Coronary IVL System is contraindicated for the following: This device is not intended for stent delivery. This device is not intended for use in carotid or cerebrovascular arteries.
Warnings— Use the IVL Generator in accordance with recommended settings as stated in the Operator’s Manual. The risk of a dissection or
perforation is increased in severely calcified lesions undergoing percutaneous treatment, including IVL. Appropriate provisional interventions should be readily available. Balloon loss of pressure was associated with a numerical increase in dissection which was not statistically significant and was not associated with MACE. Analysis indicates calcium length is a predictor of dissection and balloon loss of pressure. IVL generates mechanical pulses which may cause atrial or ventricular capture in bradycardic patients. In patients with implantable pacemakers and defibrillators, the asynchronous capture may interact with the sensing capabilities. Monitoring of the electrocardiographic rhythm and continuous arterial pressure during IVL treatment is required. In the event of clinically significant hemodynamic effects, temporarily cease delivery of IVL therapy.
Precautions— Only to be used by physicians trained in angiography and intravascular coronary procedures. Use only the recommended balloon
inflation medium. Hydrophilic coating to be wet only with normal saline or water and care must be taken with sharp objects to avoid damage to the hydrophilic coating. Appropriate anticoagulant therapy should be administered by the physician. Precaution should be taken when treating patients with previous stenting within 5mm of target lesion.
Potential adverse effects consistent with standard based cardiac interventions include– Abrupt vessel closure – Allergic reaction to contrast medium, anticoagulant and/or antithrombotic therapy-Aneurysm-Arrhythmia-Arteriovenous fistula-Bleeding complications-Cardiac tamponade or pericardial effusion-Cardiopulmonary arrest-Cerebrovascular accident (CVA)-Coronary artery/vessel occlusion, perforation, rupture or dissection-Coronary artery spasm-Death-Emboli (air, tissue, thrombus or atherosclerotic emboli)-Emergency or non-emergency coronary artery bypass surgery-Emergency or non-emergency percutaneous coronary intervention-Entry site complications-Fracture of the guide wire or failure/malfunction of any component of the device that may or may not lead to device embolism, dissection, serious injury or surgical intervention-Hematoma at the vascular access site(s)-Hemorrhage-Hypertension/Hypotension-Infection/sepsis/fever-Myocardial Infarction-Myocardial Ischemia or unstable angina-Pain-Peripheral Ischemia-Pseudoaneurysm-Renal failure/insufficiency-Restenosis of the treated coronary artery leading to revascularization-Shock/pulmonary edema-Slow flow, no reflow, or abrupt closure of coronary artery-Stroke-Thrombus-Vessel closure, abrupt-Vessel injury requiring surgical repair-Vessel dissection, perforation, rupture, or spasm.
Risks identified as related to the device and its use: Allergic/immunologic reaction to the catheter material(s) or coating-Device malfunction, failure, or balloon loss of pressure leading to device embolism, dissection, serious injury or surgical intervention-Atrial or ventricular extrasystole-Atrial or ventricular capture.
Prior to use, please reference the Instructions for Use for more information on warnings, precautions and adverse events.
https://shockwavemedical.com/IFU
Please contact your local Shockwave representative for specific country availability and refer to the Shockwave C2 Instructions for Use containing important safety information.