Category I CPT® Add-on Code Established for Coronary IVL
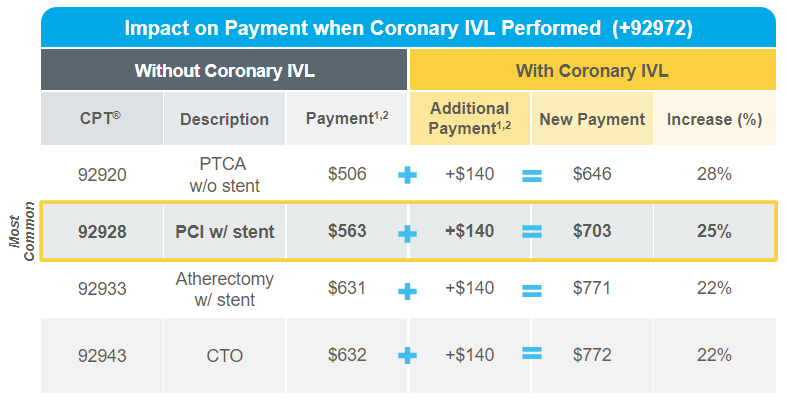
CPT® code +92972 provides an additional 2.97 Work Relative Value Units (RVU) and $140 payment for procedures involving Coronary IVL
- The Centers for Medicare & Medicaid Services (CMS) has established a Category I Current Procedural Terminology (CPT®) Add-on code for procedures involving Coronary IVL. This code is effective January 1, 2024. Prior to the establishment of +92972, there were no additional RVUs or professional fees for performing Coronary IVL.
The tables below display the value associated with CPT® code +92972 and provide examples of how utilizing the code in conjunction with a primary procedure impacts work RVUs and payment. As an add-on code, +92972 can be utilized regardless if adjunctive technologies (i.e. atherectomy, PTCA) are used in conjunction with Coronary IVL.
sssss
CPT® is a registered trademark of the American Medical Association. Copyright 1995 – 2023. All rights reserved.
1. CMS-1784-F; Medicare Physician Fee Schedule Fiscal Year 2024 Final Rule
2. Payment rates do not take into account geographical or additional adjustments. Providers should contact their local Medicare Administrative Contractor (MAC) or CMS for specific information as payment rates vary by region.
Applicable FARS/DFARS Restrictions Apply to Government Use. Fee schedules, relative value units, conversion factors and/or related components are not assigned by the AMA, are not part of CPT, and the AMA is not recommending their use. The AMA does not directly or indirectly practice medicine or dispense medical services. The AMA assumes no liability for data contained or not contained herein.
The coding, coverage, and payment information contained herein is gathered from various resources and is subject to change without notice. Shockwave Medical cannot guarantee success in obtaining third-party insurance payments. Third-party payment for medical products and services is affected by numerous factors. It is always the provider’s responsibility to determine and submit appropriate codes, charges, and modifiers for services that are rendered. Providers should select the most appropriate HCPCS code(s) with the highest level of detail to describe the service(s) rendered to the patient. Providers should contact their third-party payers for specific information on their coding, coverage, and payment policies.
Prior to use, please reference the Instructions for Use for more information on indications, contraindications, warnings, precautions and adverse events.
-
-
To see outstanding Shockwave IVL cases, follow @ShockwaveMedical on Instagram!
To keep up on the real-time Shockwave IVL activities, follow @ShockwaveIVL on Twitter
-
Coronary Important Safety Information:
In the United States: Rx only.
Indications for Use—The Shockwave Intravascular Lithotripsy (IVL) System with the Shockwave C2 Coronary IVL Catheter is indicated for lithotripsy-enabled, low-pressure balloon dilatation of severely calcified, stenotic de novo coronary arteries prior to stenting.
Contraindications—The Shockwave C2 Coronary IVL System is contraindicated for the following: This device is not intended for stent delivery. This device is not intended for use in carotid or cerebrovascular arteries.
Warnings— Use the IVL Generator in accordance with recommended settings as stated in the Operator’s Manual. The risk of a dissection or perforation is increased in severely calcified lesions undergoing percutaneous treatment, including IVL. Appropriate provisional interventions should be readily available. Balloon loss of pressure was associated with a numerical increase in dissection which was not statistically significant and was not associated with MACE. Analysis indicates calcium length is a predictor of dissection and balloon loss of pressure. IVL generates mechanical pulses which may cause atrial or ventricular capture in bradycardic patients. In patients with implantable pacemakers and defibrillators, the asynchronous capture may interact with the sensing capabilities. Monitoring of the electrocardiographic rhythm and continuous arterial pressure during IVL treatment is required. In the event of clinically significant hemodynamic effects, temporarily cease delivery of IVL therapy.
Precautions— Only to be used by physicians trained in angiography and intravascular coronary procedures. Use only the recommended balloon inflation medium. Hydrophilic coating to be wet only with normal saline or water and care must be taken with sharp objects to avoid damage to the hydrophilic coating. Appropriate anticoagulant therapy should be administered by the physician. Precaution should be taken when treating patients with previous stenting within 5mm of target lesion.
Potential adverse effects consistent with standard based cardiac interventions include– Abrupt vessel closure – Allergic reaction to contrast medium, anticoagulant and/or antithrombotic therapy-Aneurysm-Arrhythmia-Arteriovenous fistula-Bleeding complications-Cardiac tamponade or pericardial effusion-Cardiopulmonary arrest-Cerebrovascular accident (CVA)-Coronary artery/vessel occlusion, perforation, rupture or dissection-Coronary artery spasm-Death-Emboli (air, tissue, thrombus or atherosclerotic emboli)-Emergency or non-emergency coronary artery bypass surgery-Emergency or non-emergency percutaneous coronary intervention-Entry site complications-Fracture of the guide wire or failure/malfunction of any component of the device that may or may not lead to device embolism, dissection, serious injury or surgical intervention-Hematoma at the vascular access site(s)-Hemorrhage-Hypertension/Hypotension-Infection/sepsis/fever-Myocardial Infarction-Myocardial Ischemia or unstable angina-Pain-Peripheral Ischemia-Pseudoaneurysm-Renal failure/insufficiency-Restenosis of the treated coronary artery leading to revascularization-Shock/pulmonary edema-Slow flow, no reflow, or abrupt closure of coronary artery-Stroke-Thrombus-Vessel closure, abrupt-Vessel injury requiring surgical repair-Vessel dissection, perforation, rupture, or spasm.
Risks identified as related to the device and its use: Allergic/immunologic reaction to the catheter material(s) or coating-Device malfunction, failure, or balloon loss of pressure leading to device embolism, dissection, serious injury or surgical intervention-Atrial or ventricular extrasystole-Atrial or ventricular capture.
Prior to use, please reference the Instructions for Use for more information on warnings, precautions and adverse events. https://shockwavemedical.com/IFU
Please contact your local Shockwave representative for specific country availability and refer to the Shockwave C2 instructions for use containing important safety information.